Absolute zero, defined as 0 Kelvin or -273.15 degrees Celsius, is the theoretical temperature at which all classical motion of particles would cease. It presents a tantalizing concept: the possibility of completely still atoms. But does this notion hold true under scrutiny? To explore this question, one must delve into the intricate world of thermodynamics, quantum mechanics, and the behavior of matter at extreme conditions.
As temperature decreases, the kinetic energy of particles diminishes. This relationship is predominantly articulated through the kinetic theory of gases, which posits that temperature is proportional to the average kinetic energy of particles within a substance. Consequently, one might surmise that lowering the temperature would sequentially lead to a reduction in atomic motion, ultimately culminating in a state of absolute stillness at absolute zero.
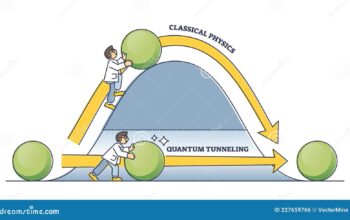
However, this simplistic interpretation of atomic behavior encounters complications upon closer examination. Quantum mechanics introduces a different paradigm of understanding atomic motion. At the quantum level, particles do not conform to the classical laws of physics. Instead, they exhibit wave-particle duality and possess intrinsic properties known as zero-point energy.
Zero-point energy is the energy that remains in a system even when it is at its lowest energy state, which is typically at absolute zero. This phenomenon derives from the Heisenberg uncertainty principle, which states that it is impossible to precisely measure both the position and momentum of a particle concurrently. In essence, even when the temperature approaches absolute zero, particles retain a minimal, but non-zero, motion due to their zero-point energy. Therefore, the notion of particles completely at rest contradicts quantum principles.
The implications of this concept extend into various realms of physics. For instance, in cryogenics, scientists have conducted experiments by cooling substances to temperatures exceedingly near absolute zero to observe quantum behaviors, such as Bose-Einstein condensates (BEC). These exotic states of matter arise when a group of bosons—particles that follow Bose-Einstein statistics—are cooled to temperatures close to absolute zero, causing them to occupy the same quantum state. Remarkably, this leads to the emergence of macroscopic quantum phenomena previously considered the domain of the atomic realm.
Furthermore, when we examine superconductivity—a state of matter that arises in certain materials at cryogenic temperatures—the properties of electrons alter dramatically. Below a critical temperature, the resistance to electrical flow disappears entirely, enabling the material to conduct electricity without loss. This occurrence exemplifies another facet where atomic motion does not cease, but instead transforms into coherent behavior at a macroscopic scale.
Moreover, when discussing atomic behavior at low temperatures, one should consider the implications of entropy and thermal equilibrium. As temperature decreases, systems tend to reach a state of minimum entropy. However, residual entropy remains an integral part of the structure, hinting at the kinetic vibrational modes retained by atoms, even at extreme low temperatures.
Ethical considerations and experimental frustrations also come into play when addressing the pursuit of absolute zero. Attaining this temperature is theoretically impossible in a physical sense, as stated by the third law of thermodynamics. Efforts to reach absolute zero continuously generate challenges, emphasizing that the closer one approaches this ideal, the more energy is required to extract any residual thermal energy from the system. Thus, while the aspiration marks a pursuit toward understanding nature, the realization of absolute zero remains an unattainable target.
In summary, the quest for absolute zero reveals layers of complexity surrounding atomic motion. The interplay between temperature and particle behavior embodies a rich tapestry woven from classical physics and quantum mechanics. Particles do not merely stop moving; rather, they persist in exhibiting a fundamental motion characterized by zero-point energy. The exploration into these realms opens gateways to innovations, pushing the boundaries of our understanding of material properties and fundamental laws of nature.
Therefore, while we may never witness atoms completely at rest, the pursuit of lowering temperatures towards absolute zero fosters deeper inquiry into the behavior of matter, enriching our scientific journey. It challenges our expectations and compels us to rethink the very nature of stillness and motion, encapsulating the essence of inquiry that lies at the heart of physics.